Wireless Technologies Drive Use of Mobile Devices in Healthcare
By Jennifer Bieksha, Bishop & Associates Inc.
Aug. 21, 2012

Advances in telehealth, wireless networks, virtual care solutions, and cloud computing are empowering healthcare providers to improve their business operations while giving them more time to provide quality care. Through telehealth, hospitals can reduce preventable hospitalizations, provide remote care for the aging population, and treat patients with chronic conditions who may have difficulty traveling. This technology can deliver new service to previously unserved markets, helping to improve care and bring in new revenue for providers.
Smartphones, tablets, and mobile apps are changing the physician-patient dynamic. They are empowering patients to take better control of managing their health, and physicians are taking advantage of the conveniences provided by these technologies. At the same time, social media is revolutionizing communication. This growing segment of healthcare-related mobile and wireless devices is now often referred to as “m-Health” (mobile health).
These dynamics have been a prime catalyst in the development of smaller, more sophisticated medical device technology for remote use, taking medical care from the hospital into the home environment. The global medical device market is predicted to grow from $322 billion in 2011 to $415 billion in 2016, despite the challenges of cost-cutting trends, the impending US medical device tax, and austerity programs in several major healthcare markets.
Medical devices used by the consumer to self-monitor their health, rather than those used in managed telehealth systems, will be the biggest opportunity for wireless technologies in healthcare over the next five years. This is the result of the slow deployment of telehealth systems, due in part to regulations  related to the use and storage of medical data. Market analysts estimate that more than 50 million wireless health monitoring devices will ship for consumer monitoring applications during the next five years, with a smaller number being used in managed telehealth systems.
related to the use and storage of medical data. Market analysts estimate that more than 50 million wireless health monitoring devices will ship for consumer monitoring applications during the next five years, with a smaller number being used in managed telehealth systems.
New wireless protocols such as Bluetooth, ZigBee, ANT+, and 4G are also fueling the growth of new applications. ANT+ is a 2.4GHz wireless network protocol used in wireless sensor networks that require low cost, low power, small form factor, and flexibility. Bluetooth low-energy technology is ideal for applications requiring episodic or periodic transfer of small amounts of data. Coin cell battery-operated sensors and actuators in medical and fitness applications now connect to Bluetooth low energy technology–enabled smart phones, tablets, or gateways.
Telehealth and the Cloud
Telehealth has transformed the practice of medicine, reducing the distance between the caregivers and those in need. Multiple telecom organizations are promoting the concept of wireless health and how it enables “access” via mobile technology such as smartphones or other wireless devices. More health organizations are starting to use remote service to help patients cut down on doctor office visits and travel time.
 Cloud service providers are able to offer cloud-based services for the healthcare industry to help monitor, analyze, and react to real-time patient information. With data coming into the cloud, the data is now more accessible, and patients want to access that data. In the past, healthcare organizations didn’t want to share their records with patients because records often contain data not strictly related to the patient’s condition. With raw data, the physicians can have their own interpretation of that data separate from the patient data. Now, patients can take all their clinical data and can carry it to any doctor throughout the system.
Cloud service providers are able to offer cloud-based services for the healthcare industry to help monitor, analyze, and react to real-time patient information. With data coming into the cloud, the data is now more accessible, and patients want to access that data. In the past, healthcare organizations didn’t want to share their records with patients because records often contain data not strictly related to the patient’s condition. With raw data, the physicians can have their own interpretation of that data separate from the patient data. Now, patients can take all their clinical data and can carry it to any doctor throughout the system.
Increasingly available health-related peripheral devices can track and upload information in real time via a wireless or wired connection to devices such as smartphones and tablets. Some of these devices are aimed at home healthcare, and patients interested in closely monitoring their health status, while others facilitate the exchange of information between patients, hospitals, clinics, and physicians. Examples include blood pressure monitors, blood glucose meters, insulin pumps, pulse oximeters, ECG monitors, portable ultrasound, and implantable devices.
Wireless technologies are also providing the aging population the opportunity to live independently with safety. Servers connected to many sensor devices can be installed at a home and monitored via wireless communications. The system can link with a blood pressure monitor, digital weight scale, motion sensor, mattress weight sensor, door sensor, pulse oximeter, or even a pair of shoes with built-in GPS. These devices are able to track and graph the day-to-day activities of the user and report any unusual pattern to the caregiver.
Mobile Health Apps
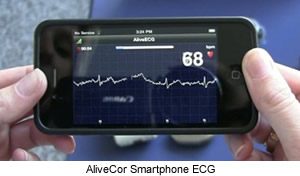
Mobile apps have dramatically changed how patients obtain information about medical conditions, along with keeping up with general wellness. There is a wealth of mobile health apps that let users transfer readings from a medical device, which can then be stored and displayed on the device or uploaded to a cloud-based system. Many consumers already use smartphone apps to track their own health and fitness results, via devices such as pedometers, speed and distance sensors, and heart-rate monitors.
Combined with new worldwide social trends that are seeing more people are interacting with technology, the acceptance of mobility has accelerated. Healthcare providers can leverage many applications to help patients better manage their health; there are more than 10,000 health applications in the iTunes store alone! Likewise, mobile apps are helping physicians further educate patients about their conditions.
Tablets are being used more and more by doctors and patients in hospitals and clinics. The iPad already has many healthcare software apps, and many medical schools are using the device for training. Portable tablets also enable doctors to interact with their patients. Fairly new, but already having a very positive impact within the medical community, are platforms that allow the physicians and patients real time together to benefit from the technology. The drawMD app allows doctors to draw on an image to show the patient exactly what he or she will be going through in surgery or to better understand a medical condition.
Miniaturization of Electronics for Medical Device Applications

The advancement of electronics in the last decade has enabled electronic design to become more compact, more portable, and easier to use. The smaller footprints of individual components, advanced manufacturing, and packaging techniques enable medical devices to be very compact. However, putting more functionality in the same amount of space, or even smaller spaces and enclosures, is an ongoing challenge for medical electronics designers.
 Some of these devices are targeted toward home healthcare and patients interested in closely monitoring their health status, while others facilitate the exchange of information between hospitals, clinics, and physicians.
Some of these devices are targeted toward home healthcare and patients interested in closely monitoring their health status, while others facilitate the exchange of information between hospitals, clinics, and physicians.
The wide range of applications for medical electronics drives unique requirements that can differ significantly from commercial and military electronics. This is particularly true for handheld, portable, and implantable medical devices that demand increased functionality with decreasing size, weight, and power (SWaP).
 Advanced packaging solutions have been successfully implemented to reduce electronics volume. The biggest challenge for connector manufacturers has not been reducing the component’s profile, but rather in reducing the printed circuit board space required by the component’s footprint. Microflex coupled with ultra-fine flip chip assembly helps to meet the challenge of extreme miniaturization and unique form factor requirements. Connector manufacturers need to reduce the pitch while offering higher signal performance at higher density. These solutions must ensure high mating cycles, low contact resistance, enhanced signal integrity, and better protection from damaging EMI and RFI interference.
Advanced packaging solutions have been successfully implemented to reduce electronics volume. The biggest challenge for connector manufacturers has not been reducing the component’s profile, but rather in reducing the printed circuit board space required by the component’s footprint. Microflex coupled with ultra-fine flip chip assembly helps to meet the challenge of extreme miniaturization and unique form factor requirements. Connector manufacturers need to reduce the pitch while offering higher signal performance at higher density. These solutions must ensure high mating cycles, low contact resistance, enhanced signal integrity, and better protection from damaging EMI and RFI interference.
What is the future of the medical device?
Mobile consumer device lifecycles are incredibly fast compared with medical device lifecycles. Mobile consumer device manufacturers typically release one or two new devices a year, and that is unlikely to change any time soon. Even if that cycle did slow down, mobile consumer devices would not get to lifecycles measured in multiple years or even tens of years, which is characteristic of the medical device world.
Medical device manufacturers must determine how to integrate the rapid lifecycle plan into the heavily regulated product development and approval process. Medical device manufacturers may soon have a consumer mobile device with built-in healthcare features. Consumer platform providers need to identify how close to the FDA line they’re going to get. The question remains:Even if the device exists, will the consumer device companies be willing to take on medical liability that comes with the device?

The medical ecosystem used to be a device, a patient, and a physician. That is changing dramatically with electronic health records, consumer platforms, and cloud computing. Wireless technologies in the medical device market will continue to expand. Telehealth, home healthcare, and increased use of smart handheld devices will drive the growth of healthcare. More devices will be connected together as reliable and secure end-to-end connections evolve. There are still issues to be resolved, but we are very close to seeing the delivery of a complete, transparent, wireless mobile healthcare system.
Bishop Notes
- The Federal Communications Commission (FCC) has voted to allocate a portion of the spectrum for connecting wireless medical devices to allow for more convenient and cost-effective health monitoring. It is referred to as the Medical Body Area Networks (MBANs). The FCC will set aside two spectrum bands; one strictly for devices used in medical facilities and a second band for remote monitoring of patients in their own homes.
- The IEEE announced two new standards and a revised standard intended to support plug-and-play, interoperable medical-device communications.
- The new IEEE 11073-00103 “Guide for Health Informatics–Personal health device communication–Overview” defines profiles for interoperable communications among personal telehealth devices used for life activity and health/wellness monitoring in domestic home, communal home and/or mobile applications and compute engines such as health appliances, set-top boxes, cell phones and personal computers.
- IEEE 11073-10103 “Standard for Health Informatics–Point-of-care medical device communication–Nomenclature–Implantable device, cardiac” defines discrete terms to be included when conveying a clinically relevant summary of the information obtained from devices such as implantable pacemakers, defibrillators, devices for cardiac-resynchronization therapy and implantable cardiac monitors.
- A revised IEEE 11073-10417 “Standard for Health Informatics–Personal health device communication–Part 10417: Device specialization–Glucose meter” has also been approved. IEEE 11073-10417 is designed to support plug-and-play interoperability among personal telehealth glucose meter devices and compute engines.
- State of the Industry: 2022-2023 Connector Sales - April 16, 2024
- Amphenol is On a Roll - April 2, 2024
- Nicomatic Proves That Two Heads are Better Than One - March 26, 2024

