Four Steps for Choosing Fluid Connectors
With so many risks in and options for connecting tubing in medical applications, it is important to have a simple and repeatable strategy for selecting the best connector solution. Here are CPC’s four tips for choosing fluid connectors.

Connections to medical equipment need to be secure and convenient while also preventing misconnections.
Choosing the right fluid connectors can improve patient safety and user convenience while maximizing sealing and flow performance. With so many risks in and options for connecting tubing in medical applications, it is important to have a simple and repeatable strategy for selecting the best connector solution. The process requires a thorough analysis of the application in order to ensure connectors will be compatible with the physical, chemical, and biological environment; be easy to use; and help prevent misconnections – whether the application involves connecting air lines for a blood pressure cuff, connecting reagent supplies to a blood analyzer, or making critical connections between a patient and a heart-lung machine. The connector selection process can be broken down into four decision-making steps.
Step One: Consider Safety Needs
Begin the connector selection process by considering the safety of the patients and healthcare professionals who will be using the connectors. Connectors need to be easy and intuitive to use so there is no danger of leaks, spills, or, in particular, misconnections. In the past, the luer was a simple, universal design that provided an acceptable connection for many applications. However, today’s escalation in the number of tubes and connections in medical laboratories, hospital rooms, and operating rooms has increased the danger of misconnections. Regulatory and reporting groups, such as the Food and Drug Administration (FDA), the Institute for Safe Medication Practices (ISMP), and The Joint Commission (TJC), have all reported patient injuries or deaths arising from misconnections.
Due to concerns over the proliferation of luer connectors, a series of new small-bore medical connector standards is being developed by a work group of the International Standards Organization (ISO) and the International Electrotechnical Commission (IEC). The ISO 80369 series of standards will define non-interchangeable connectors and will impact connector selections for a wide range of medical device applications. It is important to work with a company that understands the emerging standards and has developed products to answer misconnection concerns.
Step Two: Define the Functional Needs of the Application
The functional needs of a medical application determine the parameters for tubing and connectors. The application also dictates whether the tubing and connectors are disposable or reusable and whether valves are required. For example, many hospitals use disposable blood pressure cuffs as one means of infection control. For the manufacturer of the blood pressure monitor and cuffs, it means selecting a connector style without a valve – one that is simple, low-cost, and easy to secure and remove.
A blood analyzer is a more complex application. This application uses multiple lengths of tubing and connectors to supply the machine with various reagents or cleaning chemicals. Typically, lab technicians connect bottles of reagents to the machine via tubing with connectors on both ends. Connectors with valves are designed to eliminate spills and limit air from entering the system upon disconnect, creating a closed system and a cleaner, safer work environment. Finally, the connector and tubing materials selected need to be compatible with the reagents, solvents, and cleaners being used.
Consider the following factors as part of your decision-making process:
- Flow requirements – The inner diameter (ID) size of the tubing generally is the first consideration for the size of the connector. Pressure drops across connectors and valves vary greatly by manufacturer, and some designs exhibit less turbulence and resistance to flow than others. By comparing published CV factors for various connectors (often listed in manufacturers’ catalogs), design engineers can determine the pressure drop across the connector and match the correct size to their application requirements.
- Media effects – Connectors and O-rings need to be compatible with the media being transferred. Start with a review of published data in chemical compatibility charts to understand what materials will work acceptably with the range of chemicals, media, or reagents in the application. Remember to consider cleaning solutions and other environmental exposure. Testing of potential product materials is always a good idea to evaluate the materials’ performance in the actual application. Also, take into consideration the effects of gamma ray, electron beam, ETO, or autoclave sterilization on connector materials if the device will be sterilized.
- Temperature and pressure – Consider the range of temperatures and pressures in the application and also consider the range of temperatures the connectors might be subjected to in storage or shipping.
- Connector quality – The level of manufacturing quality of a connector dramatically affects its performance and can also play a role in aesthetics. On molded plastic couplings, parting lines (where two halves of the mold came together) and mold defects should be minimal on the connector body and absent from the first hose barb. Imperfections on barbs or threads can lead to leaks. The molded connector’s gate (the point where the plastic was injected into the mold) should be free of jagged edges that can catch or tear surgical gloves or tubing.
- Fitting type – The most popular styles of hose connection are the single- and multiple-hose barb. Single-hose barbs work well with softer tubing such as silicone rubber. However, connectors with multiple, sharp, well-made barbs, free of parting lines, provide a more secure connection over a wide range of tubing types.
- Valve options – There are many styles of valves, and their flow rates and pressure drops vary. Connectors with integral valves prevent spills upon disconnection and prevent air from entering the system. Connectors with integral precision flush-face valves are considered true “dry-break” connectors and should be selected when there is a critical need to avoid any spills, contamination, or the introduction of air upon connection.
- Mounting options – In some applications, a connector is mounted to the back, side, or front panel of a machine. Metal may be preferable for strength and a higher-end appearance; however, in most applications, plastic provides a robust, cost-effective solution.
Step Three: Consider Enhanced Connector Function
In addition to connecting tubing to facilitate and control the flow of fluid, new connector technologies can bring other functionality and performance attributes to medical devices and equipment. Intelligent connectors equipped with radio frequency identification (RFID) technology allow data exchange at the point of connection. Intelligent couplings communicate by sending RFID signals between the two separated coupling halves. Data is stored on an RFID tag embedded in the passive half of the coupling, known as the insert. An RFID reader housed in the active half of the coupling, called the body, is looking for the tag. When the two coupling halves are brought within a few centimeters of each other, the reader detects the tag, reads it, and sends the tag data to the control unit running the system. The control unit can also tell the reader to write new information to the tag. Communicating couplers can transmit information that helps protect equipment, improves processes, and even saves lives in medical applications.
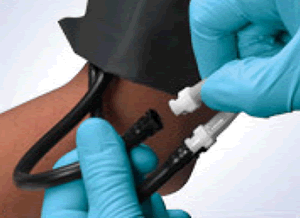
Quick connectors make single-use blood pressure cuffs affordable and convenient.
Another new connector approach that increases connector function is the hybrid quick-disconnect, which combines the transfer of power, signal, and fluids (liquids and air) in a single device. Hybrid connectors eliminate the need for multiple connections and simplify the user interface between remote tools and a device. These connectors allow technicians to quickly and safely change or replace modular tools, umbilicals, or hand pieces.
Step Four: Match Materials to the Application
The type of media flowing through a connector can influence the selection of a connector. Plastics, fluoropolymers, and alloys will all be handled differently. (For more information on specific substances and how to handle them, click the link below for the original white paper on CPC’s site.)
Although connectors are sometimes thought to be a minor component of medical equipment, they should not be relegated to an afterthought in the design process. Connectors need to be considered early in the process in order to fulfill aesthetic goals as well as product performance and patient safety objectives. Because the connector is usually the user’s primary interface with the device, it plays a key role in overall perception of the design. A well-designed connector makes the medical device easy to use and enhances the overall satisfaction with the device.
To read this white paper in its entirety, click here.
Jim Brown is the medical business unit manager at CPC.
- CNC Rotary Transfer Architecture for High-Density Connector Production - May 5, 2026
- Derating Curves for Connectors - April 28, 2026
- Smaller, Smarter, Sustainable Connectors - April 21, 2026





