Medical Interconnect Solutions Will Spur Innovation Across Markets
The COVID-19 pandemic pushed medical interconnect providers to create new strategies, capabilities, and products in 2020. In 2021, we predict that those lessons will transfer to new medical devices and care processes, and influence capabilities for other markets as well.
Connector companies are adept at aligning their capabilities with the challenging requirements of device manufacturers in a development process that ends with unique interconnect solutions that fit like puzzle pieces in new products. In 2020, companies were challenged to create new products, processes, and even new supply chains in an environment that was accelerated by the need to save lives. medical interconnect

Proactive connector companies are strengthening their strategies, technical capabilities, and product portfolios to serve what experts predict will be a long-lasting, multi-year growth spurt on the heels of the pandemic, as revitalized healthcare policies across the globe bring advanced technology into new areas of medicine. The lessons from this era will transform device design and transfer to other markets going forward.
The requirements for components used in medical devices remain highly demanding: Companies require reusable interconnect solutions that can withstand harsh environments disinfection, sterilization, and reprocessing. Components must meet stringent international standards. Solutions used in disposable equipment — used to prevent the spread of viral, bacterial, or other hospital-acquired infections — must also be low-cost. Small, lightweight, and user-friendly electronics will be essential to meet the need for portability and wearability in breakthrough products that are changing the way patient care is delivered.
End-to-End Medical Interconnect Solutions
A key trend in medical electronics is an expanding view of components as part of larger interconnect solutions. Rather than focusing solely on standard off-the-shelf connectors, many connector companies have made a noticeable turn towards custom, application-specific interconnect solutions that are vertically manufactured and integrated into value-added finished assembly solutions.
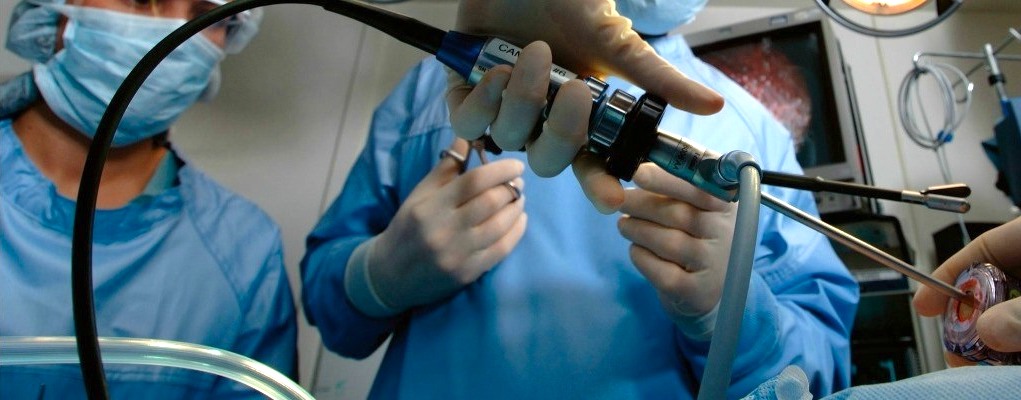
TE Connectivity’s reusable surgical assembly includes lightweight, flexible, durable cables built to withstand the sterilization processes, and proximal connectors in metal circular or customizable card-edge forms, depending on the unique requirements of the design.
TE Connectivity’s medical device solutions are now captured its Interconnect Solutions business unit, which offers connectors, wire and cable, and integrated assembly of medical electronics. The goal is to address the specific needs of a variety of diagnostic, therapeutic, and interventional medical applications.

Molex’s Hybrid Circular MT Cable and Receptacle System combines optical and electrical solutions in one connector.
Molex has reshaped its medical business from a component supplier to an original design manufacturer (ODM) end-to-end solutions provider. Phillips-Medisize is now a division of Molex called Medical and Pharmaceutical Solutions, which encompasses medical connectors, cable assemblies, and fiber technologies, along with the company’s drug delivery, diagnostics, and emerging connected health capabilities.

LEMO’s REDEL SP push-pull connectors have shock-resistant outer shells made from an FDA-approved proprietary sulfone PPSU material that enables extensive sterilization cycles and high chemical resistance. They are also available as cable assemblies equipped with Northwire’s robust, USP Class VI silicone-alternative BioCompatic cable.
LEMO, known for its plastic and metal shell circular medical connectors, as well as other specialty connectivity solutions for medical technologies, offers wire and cable products through its Northwire division. These diverse capabilities enable the company to be a full interconnect solutions provider that makes its own contacts, connectors, cable, and fully integrated cable assemblies.
Within the past five years and flying under the radar, Luxshare — a company best known for its consumer products — has also entered the medical market and quickly grown to become the fourth largest medical interconnect solutions supplier, specializing in SpO2, ECG, diagnostic ultrasound imaging, and other medical modalities.
Micro and Nano Circular Connectors
The portable and wearable medical device trend is making a dramatic impact on device designs. The desire to give patients greater comfort and flexibility, combined with the need to move some care out of clinical settings, has compelled connector suppliers to react with microminiature and nano-size circular connectors.

Microminiature circular connectors are designed to meet demanding space constraints and high-speed, lightweight, and durability requirements. Ulti-Mate offers a variety of tiny circular connectors, ranging in size from ultraminiature to near-double-density 38999 styles, and distributes through PEI-Genesis.
Rectangular Medical Interconnect Designs
To meet reduced size and weight requirements, engineers are looking for higher density, rectangular shapes that can be terminated in a planar fashion within microminiature and nano connector enclosures, and still ensure signal integrity in challenging environments. Numerous connector suppliers offering standard and custom micro-D and nano-D connectors, including AirBorn, Amphenol, Axon’ Cable, Bel Fuse (Cinch), Carlisle Interconnect Technologies, NorComp, and Omnetics Connector Corporation, to name a few.

Miniature versions of the classic D-sub connector have wide utility in medical devices. These options, available from AirBorn, NorComp, and Omnetics Connector Corporation (left to right), are designed to deliver both signal and power in a wide variety of small, portable equipment.

Another connector suitable for scaled-down applications, LEMO’s R Series uses its proven push-pull latching mechanism for a smooth and secure connection. The flat profile enables tighter front panel density for medical devices with space constraints.
Unique Capabilities, Products, and Value Propositions
Some leading medical interconnect companies have developed in-house capabilities to address the specific demands of the medical market, while others have acquired companies with differentiating capabilities and products in order to serve the needs of medical OEM customers.

Bernd Richter GmbH offers connectors for defibrillation, ECG, and other patient monitoring devices (left). CONEC offers non-magnetic D-subminiatures (right).
For example, Amphenol acquired nine companies in 2019. Two of those acquisitions, Bernd Richter GmbH and CONEC, serve the medical industry. In 2020, Amphenol also acquired Onanon for its unique medical connector technology.
Next-generation medical device designs often require a higher density of signal contacts in smaller packages. This requires significant customization to meet the electrical, mechanical, and environmental demands of the application. Some companies are designing high-density connectors into complex medical cable assemblies for electrophysiology, monitoring, therapeutics, and diagnostic imaging applications.
As the medical connector market adjusts to new ways of practicing medicine, device engineers will require strategically designed, differentiated products for these demanding applications. Look for continued innovation in this special area of the connector world.

Wayne Shockloss is the author of the Bishop & Associates’ 2020 Medical Interconnect Solutions report, which provides an in-depth review of the political, economic, sociocultural, technological, environmental, and legal (PESTEL) factors that have an impact on healthcare, medical device providers, and medical interconnect solution suppliers.
Shockloss is also the author of Top 50 Medical Interconnect Solutions Companies. This new report provides company profiles and summarizes the products, technologies, capabilities, and unique value propositions that each of the top 50 offers the medical market. Tables detail total connector sales, medical connector sales, worldwide rankings, five-year medical market forecast by subsector, as well as updated medical market expectation narratives by region and market subsectors, including the favorable COVID-19 impacts anticipated on the medical connector market.




