Multi-Functional Connectors Meet the Needs of Therapeutic Equipment
Therapeutic devices are getting smaller, smarter, serve more purposes, and are exposed to harsh cleaning and disinfection protocols. Connectors with multiple advanced features are required to meet the challenging demands of these important medical devices.
Therapeutic equipment includes a broad array of devices used to treat patients and improve their quality of life in the hospital, outpatient clinic, doctor’s office, or home. Medical device users and their patients are seeking the safest and most effective treatment, the least invasive therapies with minimal pain, quickest recovery, and the best patient outcomes.
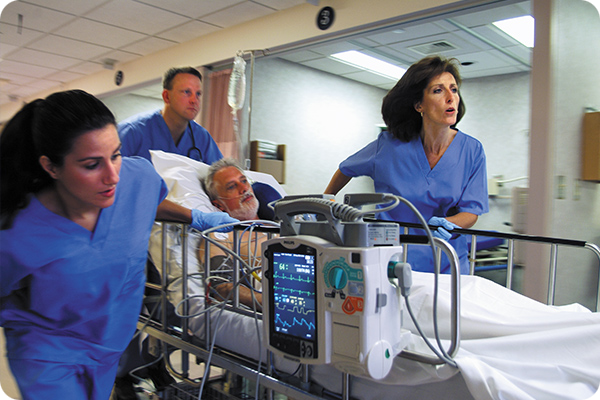
(Image courtesy of Philips Advanced Life Support Defibrillation Systems)
The coronavirus has had a negative impact on elective (non-essential) surgical procedures, which has diverted spending toward less invasive diagnostic and therapeutic treatments. There has also been a noticeable trend for quicker approvals from the U.S. Food and Drug Administration, European Medicines Evaluation Agency, and other government and regulatory bodies for innovative and breakthrough devices. These factors have favorably contributed to the growth of ventilators, point-of-care diagnostic test kits, disposable endoscopes, wellness monitoring devices, continuous glucose monitors, drug delivery devices, and many other body-worn therapeutic medical wearables.
Many of these devices gather data as part of their treatment or monitoring functions. Analysts estimate that the amount of medical data obtained by devices will increase by 100% about every 73 days in 2021, the Internet of Medical Things (IoMT) will continue to grow by more than 30% to $159 billion by 2022, and the medical device industry will grow 5.6% per year through 2024.
Many different therapeutic devices are part of this picture, including life-saving defibrillators, infusion pumps, medical lasers, ophthalmic systems, electrophysiology devices, respiratory systems, dental handpieces, and many other portable and wearable devices that require unique interconnect solutions.
Reusable connectors play a major role in these devices. Design and market factors influencing reusable connector selection for therapeutic devices include the need for high-voltage power, configurable plug-and socket-solutions, sealed push-pull connectors, user-friendly interfaces, and high-density requirements. As we review various connector designs, you’ll notice that many medical connectors fulfill multiple functions. The examples given are primarily for reusable therapeutic devices.
Factor 1: Life-saving defibrillators need locking high-voltage power connectors.
Defibrillation is a therapeutic treatment for life-threatening cardiac dysrhythmias, ventricular fibrillation, and non-perfusing ventricular tachycardia. A defibrillator delivers a controlled dose of high-voltage (360 joules) electric current to the heart to save the life of a patient in cardiac arrest. They are used to prevent or correct an arrhythmia, a heartbeat that is uneven, too slow, or too fast. As life-saving devices, defibrillator paddle connectors normally lock onto the device to prevent inadvertent disconnection.

Amphenol Alden’s Pulse-Lok auto-coupling connectors and defibrillator paddle assemblies handle high-voltage connectivity requirements.
Amphenol Alden’s Pulse-Lok connectors, available from Digi-Key, Mouser Electronics, Newark, and PEI Genesis, have an intuitive rotational mating feature that locks in place, which is ideal for life-saving defibrillator applications. Pulse-Lok connectors can be integrated into rugged cables and/or paddles for use during demanding emergency care situations.

In 2019, Amphenol acquired Berndt Richter GmbH, a manufacturer of custom ECG cables (left), and reusable locking defibrillator paddle connectors (right).

Also in 2019, GPE Group acquired NICOLAY GmbH, which offers connector systems for defibrillation and cardiopulmonary resuscitation. These connectors can withstand 12kV.
Factor 2: Small and configurable plug and socket signal solutions enable faster time-to-market.
NICOLAY GmbH offers a large variety of connector plug and socket solutions with adaptability to specific medical applications. Products are designed for extended field-life and robustness in harsh medical environments. They are optimized for high performance in fully integrated cable systems.

NICOLAY’s mini7 / mini12 Plug-In Connector System consists of a miniaturized plug and socket designed for the best possible fit to guarantee a reliable signal. These connectors offer a high dielectric strength and robustness despite a high contact density, thanks to specialty potting and sealing technology.
Factor 3: Cleanability and ease-of-use creates need for moisture-proof sealed push-pull connectors.
Plastic waterproof connectors that can withstand extreme high temperatures are ideal for challenging reusable medical cleaning and disinfection environments, including surgical, dental, therapeutic care, portable equipment, ambulance and emergency care, and patient monitoring applications.
SOURIAU-SUNBANK by EATON designs and manufactures electrical and fiber-optic connectors and interconnect systems for harsh environments. The company’s JMX Plastic Shell, JBX Metal Shell, and JDX Sealed Breakaway connector series are well-suited for therapeutic applications.

SOURIAU’s JMX plastic push-pull connectors, available from PEI-Genesis, Heilind Electronics, Air Electro, TTI Inc., and Mouser, have an audible, tactile, and visual mating recognition system that guarantees a reliable, moisture-proof, sealed, and secure connection. The JMX series is capable of over 2,000 mating cycles, is able to withstand steam autoclave sterilization per IEC 60601-1 (up to 200 cycles), and has an IP68 fluid ingress rating.

SOURIAU JBX Series of push-pull metallic connectors, available from Allied Electronics, TTI Inc., Mouser Electronics, Digi-Key, and PEI-Genesis, and are well-suited for medical home care and portable devices. Similar to the JMX series, the JBX series uses a push-pull locking mechanism that requires only two fingers for mating and has audible and visual indicators for reliable locking to more than 1,000 cycles. Available in IP40 and IP68 sealing.

SOURIAU JDX Series of sealed breakaway connectors, available from TTI Inc. and Mouser, offer a quick release breakaway system that eliminates the need for manual unlocking. It disengages at specified pull force levels to ensure a safe, efficient, and quick disconnect.
Factor 4: Therapeutic applications often need user-friendly hybrid connectors that combine features.
Therapeutic dental instruments are used to examine, diagnose, manipulate, treat, restore, and remove teeth, surrounding oral tissues, and bone structures. Dental handpieces designed for these procedures typically utilize user-friendly hybrid connectors that combine signal, power, fluidics, and pneumatics.
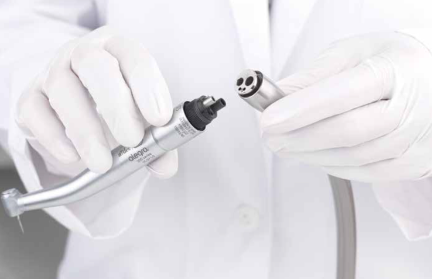
A-Dec offers electronic handpieces that are engineered for optimal power, precision, and control for handheld dental applications. The system incorporates LED lighting, ultra-quiet and low-vibration micro-motor technology with high-speed and low-speed variability, and four or five spray port options.
Caton Connector Corporation manufactures high-voltage connectors and cables for the medical and other high-reliability industries. Most of their products operate at voltages up to 100kVdc, altitudes up to 50,000 ft., and have partial discharge levels of less than 15kV RMS.

Caton QL (Quick-Locking) (left) and QL Hybrid series (right) of connectors and cables are used for medical therapeutic and surgical applications. The QL series features an auto-locking mechanism, that mates with a rotational latching mechanism that provides positive confirmation of connector engagement with an affirmative push and click. The QL hybrid series combines signal, power, pneumatic, fluidic, and fiber optics.
This customizable modular connector design allows for quick, inexpensive modifications, and is available in four shell sizes over-molded directly onto the cable of the design engineers choice. Modifications may include, custom contact inserts, strain reliefs, material, color, and marking. The Caton QL series addresses a wide range of therapeutic and dental applications that require optimal functional performance, moisture ingress resistance, cleanability, ease-of-use, and ergonomic performance.
Factor 5: OEMs require higher contact density connectors for new electrophysiology designs.
Electrophysiology procedures assess the electrical system of the heart and diagnose and treat abnormal heartbeats or arrhythmias. There is a significant amount of connector and cable assembly content used for mapping, navigation, radiofrequency (RF), and cryoablation procedures.
Many electrophysiology systems use numerous standard medical connector and cable assemblies per system, but medical device OEMs are initiating requirements for new designs that require a higher density of signal contacts in smaller packages. This requires significant customization to meet the electrical, mechanical, and environmental demands of the application. These devices are creating increased demand for smaller, higher contact density connectors.
AMETEK-TSE develops high-density interconnect solutions that provide customers with flexibility for demanding electrophysiology, and other medical applications. The DENSYTY system uses proven card edge technology to optimize high contact density in a small form factor, enhancing product performance and usability.

AMETEK DENSYTY Interconnect System can be scaled or modified to fit the needs of many applications and are tested in compliance to medical industry standards.
Therapeutic devices are getting smaller, smarter, serve more purposes, and are exposed to harsh cleaning and disinfection protocols. As a result, a growing number of connector features are required to meet the challenging demands of therapeutic applications. Connector suppliers have developed a wide range of innovative solutions to meet this evolving demand.





